Biologic purification is one of the most critical stages in therapeutic protein development. When you produce monoclonal antibodies, recombinant proteins, vaccines, or other biologics, you also generate unwanted impurities during the manufacturing process. Among the most concerning contaminants are host cell proteins (HCPs), which can affect product safety, stability, and regulatory approval.
That is why HCP antibody specificity plays such a major role in purification workflows. If your antibodies fail to recognize a broad and relevant range of host cell proteins, you risk leaving impurities behind that may compromise your final product. Understanding how antibody specificity works can help you improve process control, reduce purification risks, and strengthen analytical confidence throughout biologic development.
Understanding HCP Antibodies in Biologic Manufacturing
Host cell proteins are residual proteins released from the cells used to produce biologics. Even after purification, trace amounts of these proteins may remain in the final product. Because some HCPs can trigger immune responses or degrade therapeutic proteins, regulatory agencies expect manufacturers to monitor and minimize them carefully.
An effective HCP antibody characterization strategy for biologic purification helps you detect a broad spectrum of impurities during analytical testing. Specialized antibodies are commonly used in ELISA assays and other detection systems to measure HCP contamination levels across manufacturing stages.
When antibody specificity is poor, certain host cell proteins may escape detection entirely. This creates blind spots in your purification assessment and increases the risk of product inconsistency.
You can explore advanced analytical approaches for Hcp Antibody testing and characterization services for biologic purification workflows to improve impurity detection and process validation.
Why Antibody Specificity Is So Important
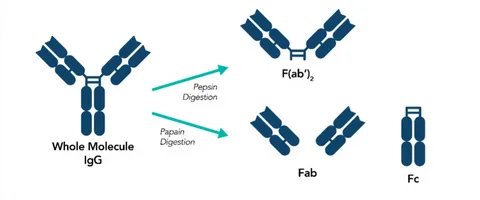
Antibody specificity refers to the ability of an antibody to recognize and bind to intended HCP targets without excessive cross-reactivity or missed proteins. In biologic purification, this specificity determines how accurately you can evaluate impurity clearance.
Improves Detection Coverage
Different host cell proteins vary in size, structure, abundance, and immunogenicity. A poorly characterized antibody may only detect a small fraction of these contaminants.
When you use highly specific and broadly reactive antibodies, you gain better visibility into residual proteins across your purification process. This allows you to identify problematic impurities before they affect product quality.
Comprehensive coverage becomes especially important when process conditions change, such as media optimization, scale-up production, or cell line modification.
Reduces False Confidence
One of the biggest risks in HCP analysis is assuming purification is effective when important contaminants remain undetected. If your antibodies miss key proteins, your analytical data may show artificially low HCP levels.
This false confidence can create major challenges later during stability testing, regulatory review, or commercial manufacturing.
Strong antibody specificity helps ensure your data accurately reflects the real impurity profile of your biologic product.
Supports Regulatory Expectations
Regulatory agencies expect manufacturers to demonstrate that their HCP detection methods provide adequate coverage of process-related impurities. They also expect scientific justification for antibody selection and validation.
Specificity studies help support:
- Method qualification
- Process validation
- Batch consistency
- Risk assessments
- Regulatory submissions
Without adequate specificity data, your purification strategy may appear incomplete during audits or filings.
The Relationship Between Purification and HCP Risk
Even highly efficient downstream purification systems cannot remove every contaminant equally. Some host cell proteins bind tightly to therapeutic proteins, chromatography resins, or filtration membranes. Others co-purify because of similar biochemical properties.
This means certain HCPs can persist through multiple purification steps despite low overall impurity levels.
Specific antibodies help you identify these difficult contaminants more effectively. Once identified, you can adjust purification conditions to target the problematic proteins directly.
For example, you may optimize:
- Chromatography conditions
- Wash buffer composition
- Resin selection
- Filtration parameters
- Process timing
Better analytical visibility leads to smarter purification decisions.
Why Generic HCP Testing Is Not Always Enough
Not all HCP assays provide the same level of sensitivity or specificity. Generic kits may fail to detect unique impurities associated with your particular host cell line or manufacturing process.
That is why many biologic developers use customized antibody approaches tailored to their production system.
You should consider factors such as:
- Host organism type
- Cell culture conditions
- Product complexity
- Purification platform
- Process scale
Customized testing often improves detection relevance and provides stronger process insight compared to one-size-fits-all assays.
How Better HCP Antibody Specificity Improves Product Quality
High-quality biologics require consistent impurity control. Better antibody specificity contributes directly to product quality by helping you:
- Detect hidden contaminants
- Improve purification efficiency
- Reduce batch variability
- Protect protein stability
- Lower immunogenicity risk
- Strengthen regulatory readiness
When your analytical tools provide reliable impurity detection, you can make data-driven process improvements earlier in development.
This proactive approach reduces expensive troubleshooting later in commercialization.
Choosing the Right Analytical Partner
HCP analysis requires technical expertise, validated methodologies, and deep understanding of protein characterization. Working with an experienced laboratory helps you interpret impurity data accurately and optimize purification strategies more effectively.
A strong analytical partner can support:
- Antibody coverage analysis
- ELISA development
- Western blot confirmation
- Orthogonal testing approaches
- Process troubleshooting
- Regulatory documentation
If you want to strengthen impurity detection and improve purification confidence, you can Contact us today for specialized HCP antibody analysis support and expert biologic testing guidance.
Conclusion
HCP antibody specificity is far more than a technical detail in biologic purification. It directly influences your ability to detect impurities, validate purification performance, and maintain product quality throughout development.
When antibody coverage is insufficient, hidden contaminants may remain undetected and create downstream risks for safety, stability, and regulatory compliance. By prioritizing highly specific and well-characterized HCP antibodies, you improve analytical accuracy and gain stronger control over your biologic manufacturing process.
As biologic therapies become increasingly complex, precise impurity monitoring will continue to play a central role in successful product development.
FAQ
What is an HCP antibody?
An HCP antibody is an antibody used to detect host cell proteins that remain during biologic manufacturing. These antibodies are commonly used in ELISA and other analytical methods to measure residual impurities.
Why are host cell proteins dangerous in biologics?
Some host cell proteins may trigger immune responses, reduce drug stability, or interfere with therapeutic performance if not adequately removed during purification.
How does antibody specificity affect HCP testing?
Higher specificity improves impurity detection accuracy by identifying a broader and more relevant range of host cell proteins within the manufacturing process.
Why do biologic manufacturers perform HCP coverage analysis?
Coverage analysis helps determine whether HCP antibodies can recognize enough relevant impurities to support reliable purification monitoring and regulatory expectations.
Can purification remove all host cell proteins?
No purification system removes every host cell protein completely. Certain impurities may persist because they co-purify with the therapeutic product or bind strongly to purification materials.
Why is customized HCP testing important?
Customized testing improves detection relevance for specific host cell lines and manufacturing conditions, leading to more accurate impurity monitoring and process optimization.