Endotoxin contamination remains a common but often underestimated issue in plasmid DNA preparation, particularly for applications involving mammalian cell transfection and in vivo studies. Even trace levels of endotoxins can trigger inflammatory responses, reduce cell viability, and significantly compromise transfection efficiency. As a result, removing endotoxin from plasmid DNA has become a critical step in ensuring reliable experimental outcomes.
However, effective endo toxin removal is not simply a matter of purification. Many commonly used approaches introduce trade-offs, including DNA loss, structural damage, or inconsistent results. Understanding these challenges is essential for selecting the right strategy.
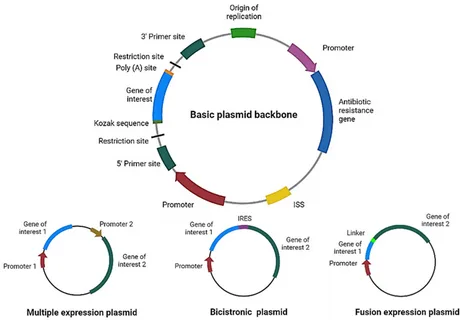
Sources of Endotoxin in Plasmid DNA Preparation
Endotoxins are lipopolysaccharides (LPS) derived from the outer membrane of Gram-negative bacteria such as E. coli, which is widely used for plasmid amplification. During cell lysis and DNA extraction, these are released and can co-purify with plasmid DNA due to their physicochemical properties.
Their amphiphilic nature allows them to form stable complexes with nucleic acids, making complete removal difficult. Without proper control, it’s contamination can persist even after standard purification workflows.
Why Endotoxin Removal Matters for Downstream Applications
The presence of endo toxins can directly interfere with biological systems, particularly in sensitive applications. In mammalian cell culture, these can activate immune signaling pathways, leading to altered gene expression and reduced cell viability. In transfection experiments, this often results in lower efficiency and inconsistent performance across replicates.
More importantly, it’s contamination can introduce variability that is difficult to detect or control, especially in comparative studies or multi-batch experiments. This makes reproducibility a significant concern when using non-optimized plasmid preparations.
- Reduced transfection efficiency due to cellular stress responses
- Increased variability in gene expression and functional readouts
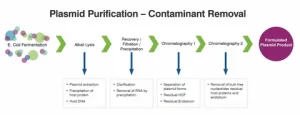
Common Strategies for Endotoxin Removal
Several methods are used to reduce it’s levels in plasmid DNA preparations. These approaches differ in mechanism, efficiency, and impact on DNA integrity.
Detergent-Based Removal
Detergents such as Triton X-114 are commonly used to separate it through phase partitioning. This method can be effective but requires careful optimization, as incomplete phase separation or residual detergent can affect downstream applications.
Affinity-Based Methods
Affinity resins designed to bind endotoxins provide a more targeted approach. These systems rely on interactions between it and specific ligands, allowing selective removal. While relatively efficient, their performance may vary depending on sample composition and binding capacity.
Chromatography-Based Purification
Chromatographic techniques, including ion exchange and size exclusion, are widely used for plasmid purification and play an important role in endotoxin removal strategies. These methods offer better scalability and control but often require optimized conditions to achieve low endotoxin levels without affecting DNA yield.
Phase Separation Techniques
Organic extraction or two-phase separation methods can help remove it based on their solubility properties. However, these approaches can be labor-intensive and may introduce variability if not carefully controlled.
Comparison of Endotoxin Removal Methods
| Method | Principle | Advantages | Limitations |
| Detergent-based | Phase separation | Simple and cost-effective | Residual detergent, variable efficiency |
| Affinity resins | Selective binding | High specificity | Limited capacity, cost |
| Chromatography | Charge/size separation | Scalable, reproducible | Requires optimization |
| Phase separation | Solubility differences | Effective in some cases | Labor-intensive, variable |
One of the most important challenges in it’s removal is maintaining plasmid DNA yield and integrity. Aggressive purification methods can lead to DNA loss, shearing, or conformational changes that affect transfection performance.
This trade-off is particularly critical in applications requiring large quantities of high-quality DNA, such as gene therapy research or in vivo studies. In these cases, achieving both low endotoxin levels and high DNA recovery is essential but technically demanding.
Toward More Reliable Endotoxin-Free Plasmid Preparation
As experimental systems become more sensitive and regulatory expectations increase, the demand for consistent and endotoxin-free plasmid DNA continues to grow. Rather than relying solely on post-extraction cleanup, many workflows now emphasize controlled production processes that minimize endotoxin contamination from the start. This proactive approach ensures greater consistency, reduces the need for costly purification steps, and improves the overall reliability of the plasmid DNA, which is crucial for high-stakes applications such as gene therapy and in vivo studies.
This approach typically involves optimized bacterial culture conditions, refined purification strategies, and rigorous quality control to ensure it’s levels remain within acceptable limits while preserving DNA quality.
Conclusion
Removing it from plasmid DNA is a critical but complex process that directly impacts the reliability of downstream applications. While multiple strategies are available, each comes with limitations that can affect DNA yield, integrity, and reproducibility.
A more integrated approach that combines controlled production with optimized purification can provide a more reliable solution, particularly for applications requiring high-quality, endotoxin-free plasmid DNA. By refining both the production process and purification techniques, researchers can achieve better consistency and minimize variability. As research and therapeutic applications continue to evolve, ensuring both low endotoxin levels and consistent DNA performance will remain a key priority in plasmid preparation workflows. This approach not only enhances reliability but also ensures the scalability and reproducibility needed for clinical and experimental success.